#food supplement #food supplement filing #health care #SAMR #production process research report #product formula #hygiene #technical requirements #quality requirements
Explanation:
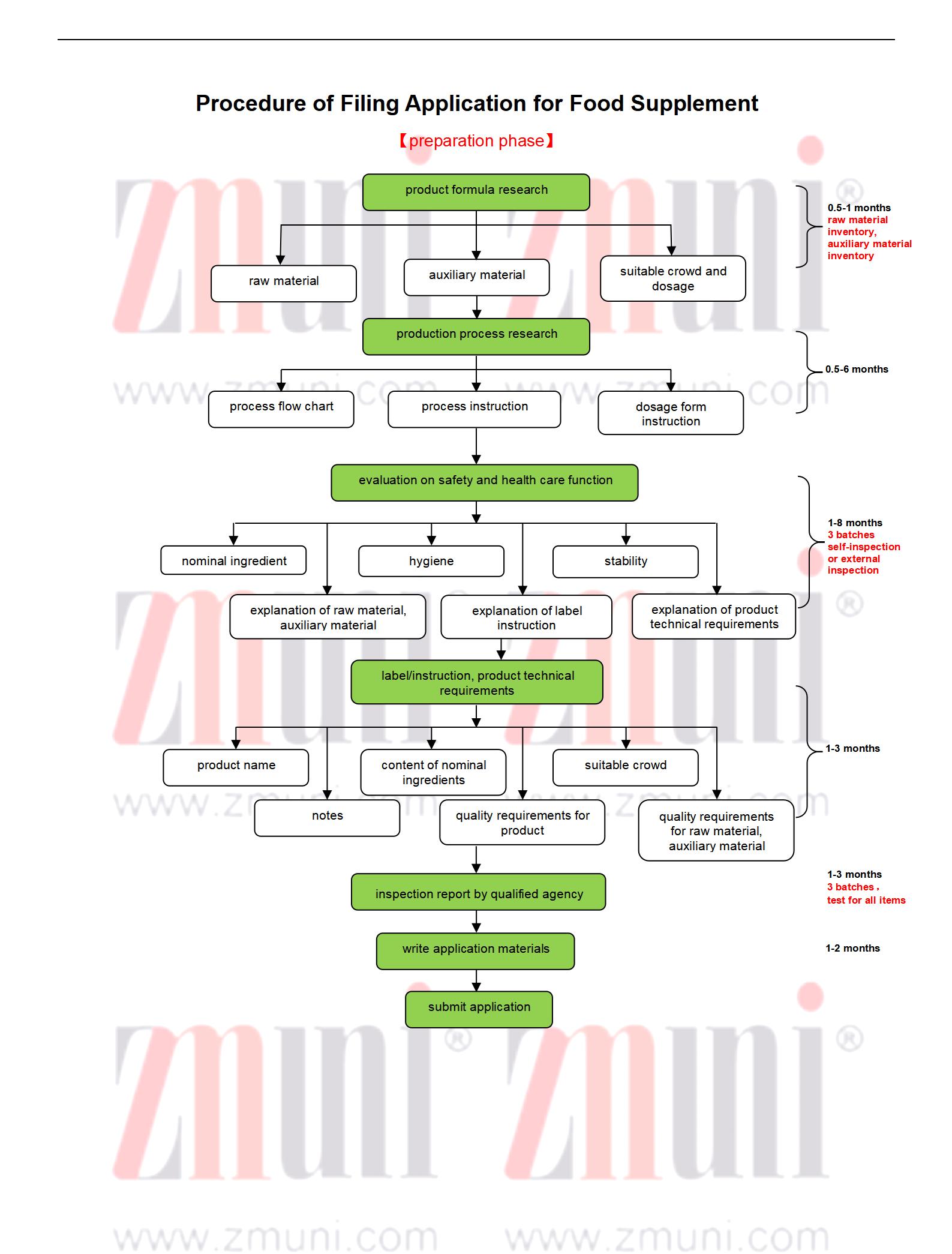
*Product formula research: The key is that it should comply with raw material inventory, auxiliary material inventory
*Production process research: The key is that it should comply with the requirements for production process and dosage form prescribed in the regulations
*Safety evaluation: The key is the nominal /efficacy ingredients, hygiene, and stability inspection report. Enterprises can self-inspect.
*Inspection report by qualified agency: This external inspection is necessary. The inspection agency or the batch of products of it should be different from those of safety external inspection(if any).
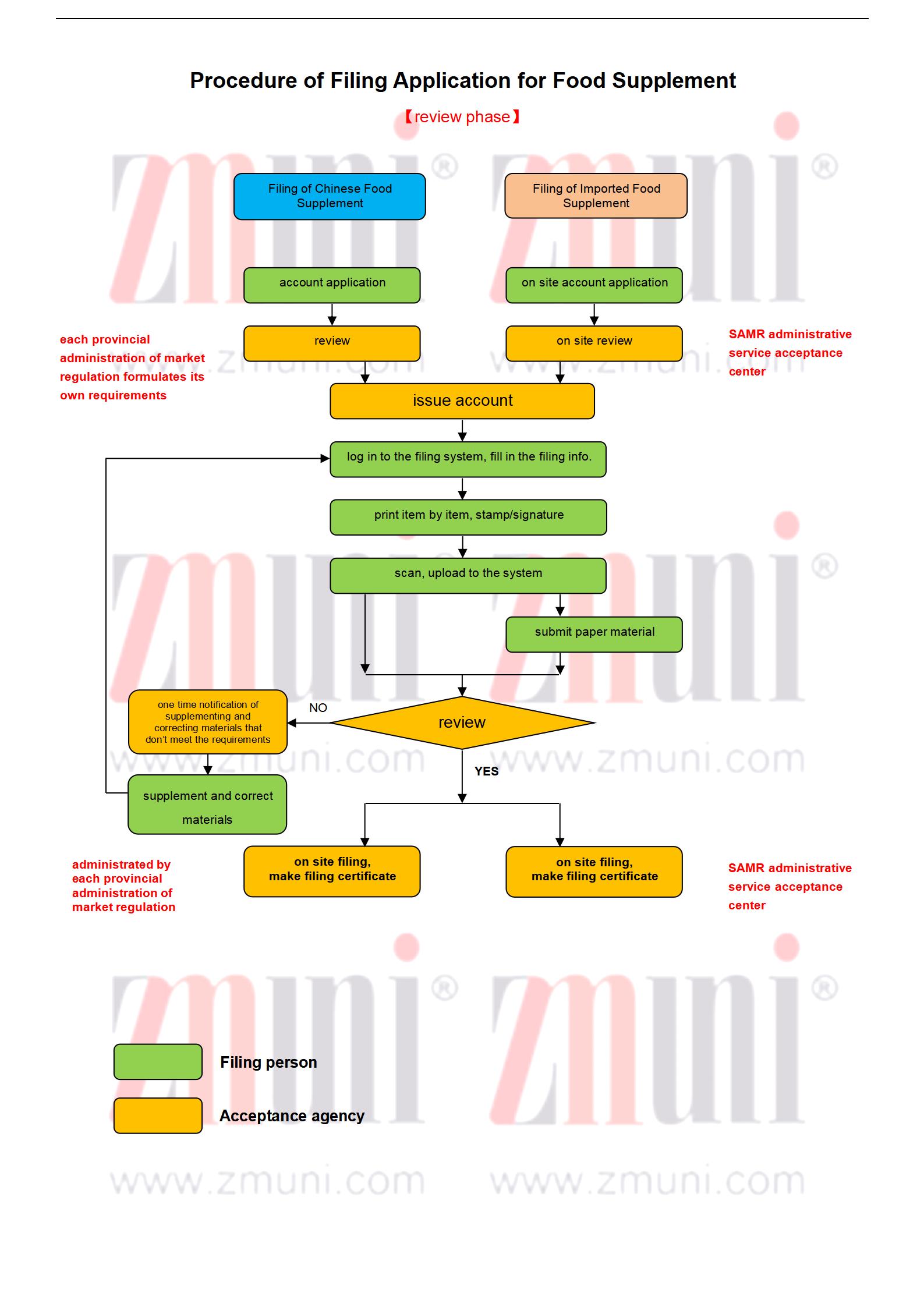
*Import registration: The overseas applicant shall designate its branch within China or authorize the main company within China to submit the application and act as a Chinese contact.
————————————






0 Comments